– Spotlight on the Orthopedic Imaging at UMC Utrecht, the Netherlands –
– Prof. Harrie Weinans & Dr. Bart van der Wal –
Metabolic imaging of osteoarthritis
Dr. Harrie Weinans is Professor of Tissue Biomechanics and Implants at TU Delft & UMC Utrecht, Department of Orthopedics. He specifically focuses on Osteoarthritis (OA), a form of progressive arthritis. Despite this disease’s large and growing worldwide burden, many pharmaceutical companies have reduced or abandoned drug development. There are currently no reliable ways of measuring whether a specific treatment is working or not. Early detection and evaluation of OA’s course are therefore crucial. Prof. Weinans has become one of the European project coordinators focused on metabolic imaging for better diagnosis and treatment in OA (1).
Dr. Weinans, it has been reported that most OA animal models are not entirely representative of naturally occurring OA as observed in humans (2). Can you comment on the animal models you find best suited to perform your OA research studies?
Because of the ultra-high resolution of both the X-ray CT and SPECT modalities of the MILabs U-SPECT/CT system, we can focus on small rodent models, mice, and rats. Since the occurrence of spontaneous OA in animals is rare and disease progression occurs at a slow rate and a specific genetic predisposition often underlies such models, we prefer experimentally induced OA. For mice, we typically inject biochemical compounds in the joint such as mono-iodoacetate (MIA) or collagenase and use saline in the opposite joint as a control. For rats, we can use surgical models such as the Groove OA model where the articular cartilage is damaged locally without damaging the underlying subchondral bone, or biomechanical models that use a motorized rat treadmill that result in minor to moderate OA related cartilage lesions.
In a recent webinar, you stated that the cause of osteoarthritis is an unresolved question as to which of two things causes the other: either wear and tear causing inflammation, or is it inflammation causing the degeneration of tissues with mechanical problems in the joint as a consequence?
Yes, this is the typical “chicken or egg” issue. Nobody really knows. However, it is true that for osteoarthritis “mechanics meets immunology”. Hence both ultra-high resolution in vivo anatomical X-ray CT and metabolic SPECT imaging capabilities of the employed SPECT/CT is of crucial importance. With high-resolution CT, we can see for instance perforations in the subchondral plate that otherwise would require histology work. During all our work, we have always observed the same phenomenon of subchondral perforations no matter what OA model was used. Unfortunately, these structural changes are most often only clearly visible in end-stage OA, in other words when the disease has already progressed significantly. With ultra-high-resolution SPECT, on the other hand, we are aiming at visualizing biochemical markers to detect OA in an early stage or to predict disease progression. Because MILabs SPECT technology has demonstrated that it can detect bone turnover in small mice using 99mTc-MDP with great precision, we feel that it is a great tool to eventually find biomarkers for the early detection of osteoarthritis and monitor disease progression.
Can you briefly comment on the current progress you have made towards finding biomarkers to identify early OA changes, both systemic- and locally within a joint?
We have used U-SPECT/CT imaging to visualize folate receptor β which is expressed in activated macrophages and can be targeted with a 111In-labeled folate tracer.. A more recently developed folate conjugate with albumin binding entity has been tested in the previously described OA Groove model (3). Using this rat Groove model in combination with a high-fat (HF) diet enabled us to study the contribution of obesity related metabolic OA, especially in its early phases. We feel this is a very relevant model, as obesity and metabolic dysregulation are an increasing concern in the western population. Using this model (see illustration below), we could visualize activated macrophages in vivo, as a measure for disease activity and study the role of these macrophages in the disease process of OA. The presence of activated macrophages by SPECT/CT was additionally confirmed by immunohistochemistry.
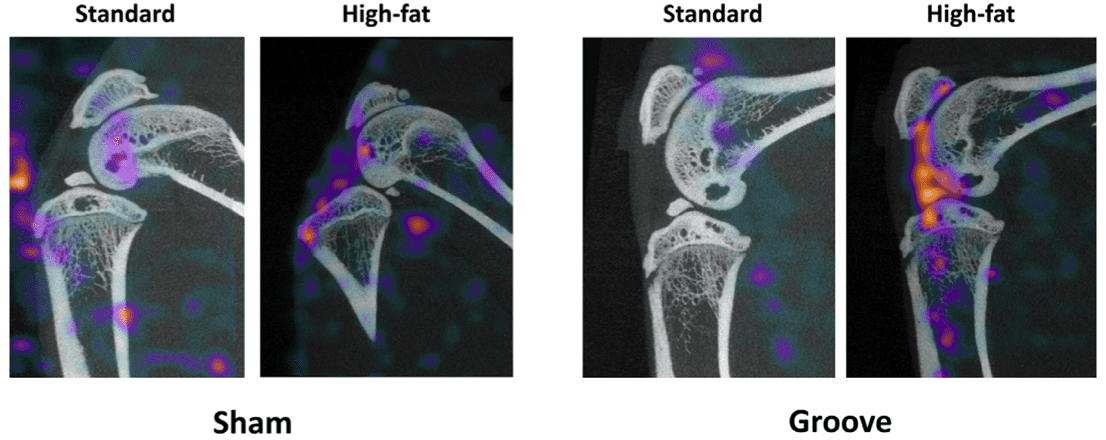
Representative MILabs SPECT/CT reconstructions of sham-operated control knee joints (left) and the mechanically-induced cartilage damage (right), 24 hours after injection of folate radio-conjugates for both conditions. Increased SPECT activity is clearly observed in the experimental grooved knee joints compared to the sham-operated controls.
Click here for Prof. Weinans video-lecture on metabolic osteoarthritis imaging
Radiotherapy imaging of implant infections
Dr. Bart van der Wal, MD, Ph.D., Orthopedic Surgeon at UMC Utrecht research is focused on the development of radioimmunotherapy (RIT) for non-invasive treatment of implant associated infections and closely collaborates with Harrie Weinans on the development of Radionuclide therapy of implant infections

Dr. van der Wal, what is your rationale for developing RIT as an alternative treatment for implant-associated infections?
Prosthetic joint infection (PJI) is a devastating complication after a joint replacement. PJI and its treatment have a high monetary cost, morbidity, and mortality. PJI is the leading cause of failure for knee replacements and the third leading cause of failure in hip replacement. The lack of success treating PJI with conventional antibiotics alone is related to the presence of bacterial biofilm on medical implants. Consequently, surgical removal of the implant and prolonged intravenous antibiotics to eradicate the infection is necessary prior to and after implanting a new prosthetic joint.
Through in-vitro work (4), we had already demonstrated the efficacy of radio-immunotherapy using alpha-RIT with 213Bi monoclonal planktonic cells and biofilms of Methicillin-resistant staphylococcus aureus (MRSA). With MILabs U-SPECT/CT in vivo imaging technology, we recently demonstrated that the use of radiolabeled specific antibodies, which target wall teichoic acids, adhere to the biofilm and the next step will be a therapeutic study to investigate if they can eradicate planktonic MRSA and biofilm on rodent implants.
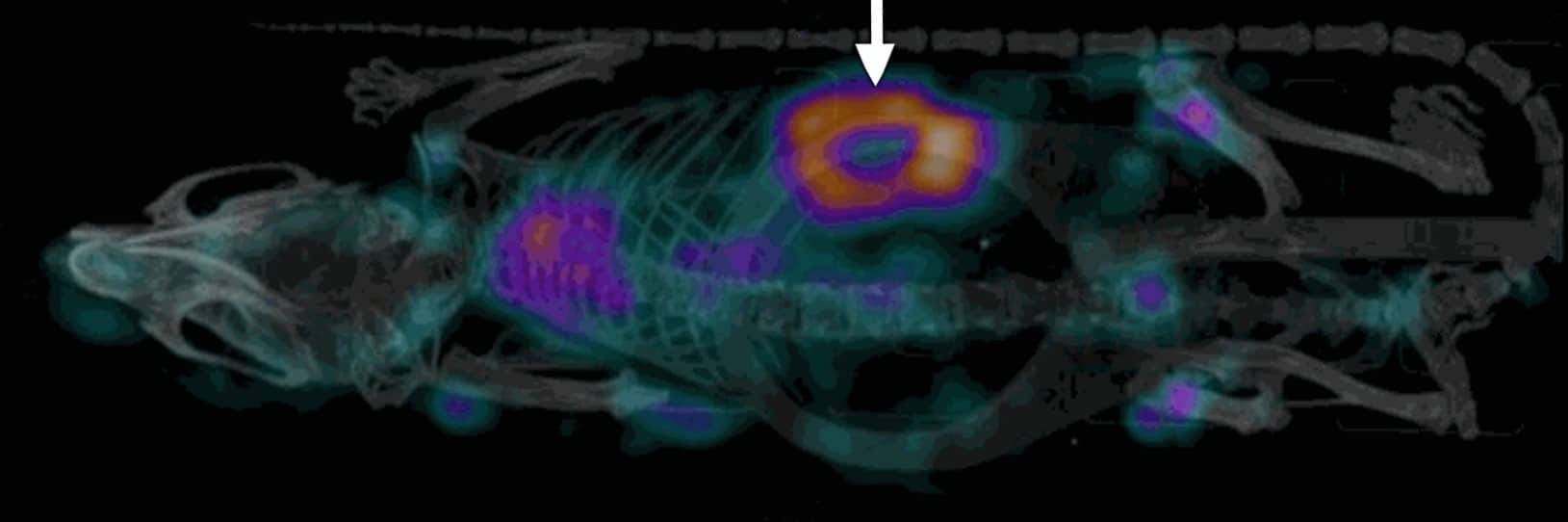
Mouse SPECT of Wall Teichoic Acid antibody against Staphylococcus aureus
labeled with Indium 111, showing clearly the S-aureus infection
Click here for Dr. van der Wal’s video-lecture on Radionuclide Therapy of Implant Infections
(1) Metajoint: H2020-EU.1.2.1. Grant agreement ID945993
(2) E. Teeple et al., Animal models of osteoarthritis: challenges of model selection and analysis. AAPS J. 2013; 15(2)
(3) HM de Visser et al., Imaging of Folate Receptor Expressing Macrophages in the Rat Groove Model of Osteoarthritis: Using a New DOTA-Folate Conjugate. Cartilage. 2018 Apr.
(4) B. van Dijk et al., Radioimmunotherapy of methicillin-resistant Staphylococcus aureus in planktonic state and biofilms. PLoS ONE 15(5): e0233086. PLoS ONE 15(5): e0233086.
