Spotlight on Sandra Heskamp, PhD: At the forefront of immunotherapy and cancer treatment research
Dr. Sandra Heskamp is an acclaimed researcher at the Radboud Institute for Molecular Life Sciences. As associate professor, she performs pioneering research in the Radiology & Nuclear Medicine department with a focus on immunotherapy and cancer treatment.
Over thirty-five scientists at the institute employ MILabs’ SPECT and CT imaging in their research. Using microSPECT/CT, Dr. Heskamp published the first preclinical study ever showing the feasibility of noninvasive in vivo imaging of PD-L1 expression in tumors. This landmark imaging study on immune checkpoint inhibition, is one of the most-referenced articles in the field of anti-PD-1:PD-L1, a research area that according to ResearchGate 2018 is clearly dominating what immunotherapy researchers are reading today.
Your team was one of the first to provide evidence that noninvasive imaging of PD-L1 in tumors with a mAb-tracer is technically possible. Can you elaborate on the imaging instrumentation challenges you encountered?
“The biggest challenge is in making immune checkpoints, immune cells, and their heterogeneous distribution in the tumor microenvironment visible. The expression levels are low and the number of immune cells is small, resulting in low signals. Our MILabs U-SPECT/CT system features high resolution imaging and provides excellent resolution/sensitivity trade-off for nuclear molecular imaging. Although in general a SPECT system has a lower sensitivity than a PET system, for preclinical imaging in mice, a conventional PET system cannot offer the resolution required to assess the heterogeneity of PD-1:PD-L1 in the tumor environment. With the help of MILabs, we were able to optimize sensitivity while maintaining the required resolution to noninvasively determine PD-L1 expression and accessibility of various tumor lesions with radiolabeled anti–PD-L1 antibodies.” (Go deeper: https://cancerres.aacrjournals.org/content/75/14/2928)
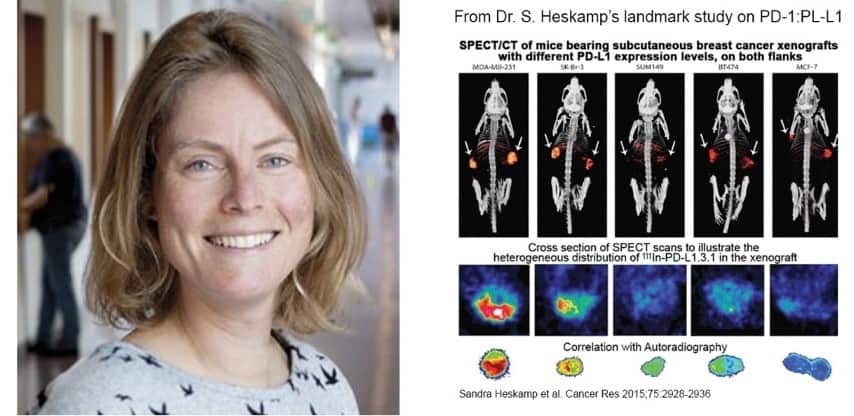
How important is the ability to image PD-L1 expression within the tumor microenvironment?
“While programmed death ligand 1 (PD-L1) expression in tumors seems to be a prerequisite for treatment response, it is heterogeneously expressed within tumor lesions and can change upon disease progression and treatment. This is clinically relevant, since it accounts for immunotherapy target accessibility. Several factors such as blood vessel density, vascular volume, vascular permeability, and interstitial fluid pressure, determine whether antibodies can reach the tumor site and bind to its target. In preclinical imaging research, the high-resolution of microSPECT/CT enables such target assessment studies.” (Go deeper: https://cancerimmunolres.aacrjournals.org/content/7/1/150)
Are there specific MILabs imaging system properties that would be useful to further advance your research?
“As documented in a recent scientific publication on PSMA-targeting agents for radio- and fluorescence-guided prostate cancer surgery, the combination of near-infrared (NIR) fluorescence imaging with nuclear imaging can be of additional value for accurate staging and complete removal of tumor tissue. In this setting, radionuclide detection allows for pre-operative visualization of the tumor lesions by SPECT imaging and sensitive intraoperative localization of tumor lesions with a gamma probe, while fluorescence imaging enables real-time tumor visualization and delineation. Preclinical SPECT imaging is a valuable preclinical tool for the development of these multimodal tumor-targeting agents.” (Go deeper: http://www.thno.org/v09p6824.htm)
How about your recent work as published in Nature on targeted alpha therapy (TAT)?
“We understand that MILabs has developed high-energy collimators for its microSPECT system, with the ability to image alpha emitters at sub-mm resolution in mouse tumor models. We encourage this instrumentation development since we have an active TAT research program with a focus on the use of 225Ac tracers. We are currently investigating the use of polymersomes as nanocarriers for 225Ac to retain harmful recoil nuclides (such as 213Bi) in alpha radionuclide therapy.” (Go deeper: https://www.nature.com/articles/s41598-019-48298-8)
You are connected to the Rare Cancer research theme of the Radboud Medical Center?
“I have a particular interest in rare cancers because they are characterized by poorer survival rates than more frequently observed cancers. As a consequence, there is often a delay in correctly diagnosing while fewer adequate therapies are available. Moreover, there is a lack of preclinical models (cell lines, organoids, patient-derived xenografts) for basic and translational research. Our current focus is on using U-SPECT/CT for the development of new tracers for quantitative imaging of the hypoxia-related marker CAIX expression in head and neck cancers. High-resolution microSPECT/CT enables us to assess the distribution of hypoxia as a radio-resistance feature within the tumors. The Radboud University Medical Center is well equipped to handle the challenges of rare cancer research since it maintains several patient-derived xenograft models.” (Go deeper: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6364270/)
Can you tell us more about the Bas Mulder & VENI awards you won?
“In 2016 I won both awards. The most rewarding outcome for me was going to Alpe D’Huez-France as winner of the Bas Mulder award. Every year, a group of cyclists go up and down that mountain in France to collect money for cancer research. The Dutch Cancer Organization (KWF), as sponsors of the Bas Mulder award, invited me to present my work to those volunteers. As an avid cyclist I participated in this event myself and it was a great experience for me to return to Alpe D’Huez to talk about my research to the people that go through all that hardship to fund it.”
