Professor Vesna Sossi, Ph.D. is blazing a trail with her theranostic research program at the University of British Columbia (UBC) and TRIUMF Canada’s Particle Accelerator Centre. As the director of the UBC Positron Emission Tomography (PET) Imaging Laboratory, Dr. Sossi and her colleagues are using the MILabs VECTor PET/SPECT/CT system to characterize and image isotopes in a way that was previously impossible, to help develop completely new cancer therapies.
Tell us about your theranostic research program:
The theranostic research program is spearheaded by research at TRIUMF, which is very unique because it can make some of the alpha-emitting radiotherapy isotopes that are really difficult to produce. Jason Crawford, Andrew Robertson, Tom Ruth and Paul Schaffer launched and worked on the program to create the radioactive beams to produce actinium and astatine1. My involvement in the program is primarily through imaging. We did find that the VECTor/CT system is extremely well suited for this type of work. We couldn’t have done imaging of these isotopes without the unique capabilities that VECTor offers.
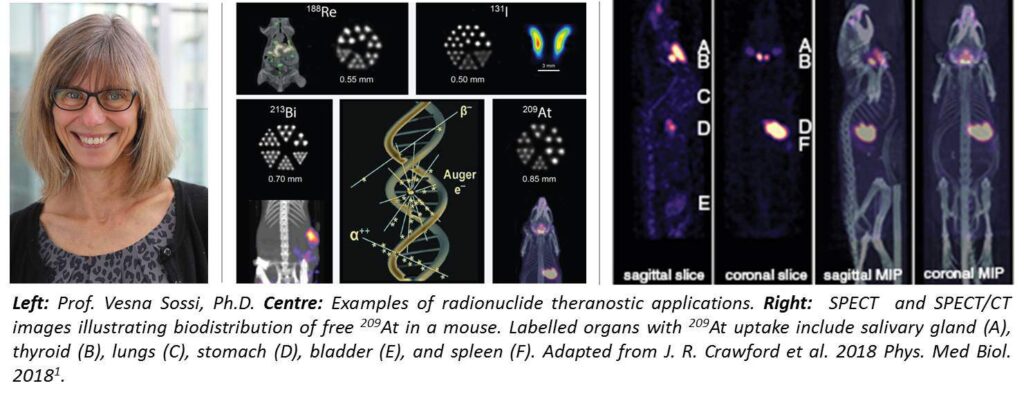
The interesting difficulty is that the amounts of these alpha emitters that are being produced are very low. In terms of imaging, these alpha emitters tend to produce, through the decay cascade, several gamma rays of various energies requiring sensitive equipment for detection. It’s important to be able to isolate or identify the optimum gammas or the gammas with the optimum energy for imaging. That required quite a lot of very careful image evaluation and different types of reconstruction.
How did the MILabs VECTor PET/SPECT/CT support you in the specific challenges with imaging these isotopes?
Because we are researching the feasibility of imaging radionuclides that emit gammas at several different high energies simultaneously, we feel this can only be done with VECTor. The unique property of this system is that we can really look at gammas with such a high range of energies. The gammas that originate from astatine covers quite a large range of energy from let’s say 70 to 92 keV x-rays, to high energy decay products of 790 keV (which is one of the gammas). You’re really exposing the instrument to a very wide range of energies. In fact, one of our first points was trying to find the best collimator for it. The low energy collimators, in principle, would have been better, but there was so much contamination from the energy gammas that we ended up finding that a high energy collimator was better. The ability to detect all of the different energy peaks with different collimators is a unique property of VECTor that we took advantage of in doing this work. Other systems may also be able to detect gammas of different energies, but unless you have the special cluster pinhole collimator2, the higher energy gammas are going to entirely smear your results because of contamination with down-scatter and collimator penetration. What is important is the combination of good energy resolutions and special collimators that the VECTor system has available.
What are other features from the latest MILabs systems that used to accelerate your theranostic research program?
It’s really the ability to image many different gammas at separate different energies3. With a traditional PET system, of course, you cannot do that because all you are doing is detecting 511 keV photons in coincidence. With a traditional SPECT system, on the other hand, you would have lots of troubles with high energy gamma rays. They wouldn’t be as well suited for this type of research as the VECTor is.
We also do fast dynamic SPECT for several studies because we are interested in the drug uptake pattern and this can only be done using VECTor fast dynamic SPECT capabilities.
What is your opinion on how alpha and/or beta radiotherapy could impact the treatment of cancer?
The advantage of alpha emitters is that the range of alpha emission is just really a few cells. If you want a very targeted and localized therapy, that’s already advantageous. Beta emitters have longer ranges of a millimeter of two or so, and therefore they really reach a relatively larger radiation area.
It’s really dependent on the type of target whether you would choose one over the other. One of the problems with alpha therapy is that many of these alpha emitters have a decay chain where there’s not a single alpha emission, but rather two or three. Therefore, you have to make sure that this doesn’t cause the breakage between the alpha and the biomolecule the alpha emitter is attached to because it’s that biochemical that gives the specificity to target various cancers. If that bond breaks, then the alphas can just go anywhere and potentially damage other areas. Ensuring that there is no such break is an important issue in for potential cancer therapies that are still under research and can be accessed with imaging at a wide range of gamma energies.
What are your other research plans for the future?
My team has diverse research plans such as: studies with gold-coated nanoparticles labelled with alpha or gamma emitting isotopes as potential theranostic agents, characterization of 225Ac labelled targeted alpha therapy agents through 213Bi and 221Fr imaging for drug delivery4, pharmacokinetic assessments of novel immunotherapeutic agents5, 118Re- labelled microspheres as potential cancer therapeutic agents6, optimization of dual-isotope imaging protocols, multi-tracer imaging in rodent models of dementia and traumatic brain injury, and the determination of the feasibility of imaging with 11C. We also want to do brain imaging in mice with VECTor. We do have a PET scanner, but the resolution is not as good as with the VECTor.
We will continue to do imaging of alpha emitters, especially with actinium, for example. The actinium decays in a way that you get francium and bismuth, which are themselves alpha emitters. That work has just started and the VECTor is a very, very well-suited instrument.
References:
1. J.R. Crawford, et al. Evaluation of 209At as a theranostic isotope for 211At radiopharmaceutical development using high-energy SPECT. Phys Med Biol. Vol: 63(4): 04502, January 2018.
2. Walker MD, et al. Performance assessment of a preclinical PET scanner with pinhole collimation by comparison to a coincidence-based small-animal PET scanner. Journal of Nuclear Medicine, Vol 55(8): 1368-1374, June 2014.
3. Sobol N, et al. Synthesis and targeting of gold-coated 177Lu-containing lanthanide phosphate nanoparticles—A potential theranostic agent for pulmonary metastatic disease. APL Bioengineering. Vol 2(1): 016101, December 2017.
4. Robertson K, et al. Multi-isotope SPECT imaging of the 225Ac decay chain: feasibility studies. Phys Med Biol. Vol 62: 4406–4420. May 2017.
5. Schmitt V, et al. Quantitative SPECT imaging and biodistribution point to molecular weight independent tumor uptake for some long-circulating polymer nanocarriers. RSC Advances. Vol 8: 5586-5595. February 2018.
6. Esquinas PL, et al. 188Re image performance assessment using small animal multi-pinhole SPECT/PET/CT system. European Journal of Medical Physics. Vol 33: 26–37. January 2017.

