Translational Nanobody research by In-vivo Cellular and Molecular Imaging (ICMI)
Tony Lahoutte, PhD. MD is head of the Nuclear Medicine Department of the University Hospital Brussel and principal investigator at ICMI at the Vrije Universiteit Brussel, Belgium.
Can you explain what the research focus is of your group in Brussels?
Twenty years ago, Prof. Casterman-Hamers at the VUB discovered that antibodies derived from Camelidae have unique properties such as a single domain antibody fragment of only 15 kDa in size with very high specificity. Because of the very small size – ten times smaller than human monoclonal antibodies – these antibody fragments, called Nanobodies, diffuse very quickly to the target and unbound nanobodies are rapidly excreted from the body. We study the stability of Nanobodies, the ability to produce these in large quantities and the flexibility to modify and label these probes with a wide variety of radio isotopes. For instance, labelling with i.e. 111In, 99mTc, 18F, 68Ga and 177Lu, may render Nanobodies ideal for future targeted nuclear diagnostic and therapy strategies. Our research is focussed around oncology, cardiovascular disease, and inflammation. In our research high throughput in vivo screening based on imaging is equally important as in vitro screening of new potential imaging and therapeutic probes. Moreover, we aim to translate potential candidates rapidly into phase 1 and 2 clinical trials.
Your MILabs scanner has the unique capability to image both SPECT and PET in the same animal. Why is this so important for your research?
Because we are interested in a wide variety of Nanobodies, each with their own preferred labeling strategy, we want to have full flexibility to image and quantify Nanobody uptake in vivo. Currently, we use radioisotopes with a short half time such as 99mTc, 68Ga and 18F because diffusion and excretion pathways of our Nanobodies are short, within 1 to 3 hours after injection. Moreover, the ability to image simultaneous SPECT and PET tracers in the same animal at exactly the same time gives us unique information on certain combinations while providing maximum flexibility. For example, if we decide to change from a preclinical SPECT labeled Nanobody to a PET labeled Nanobody for clinical phase 1 trials, we can easily compare these tracers one-on-one in vivo with the VECTor system. Additionally, the high-energy collimators also allow us to test the targeting of therapeutic isotopes such as 131I.
Can you explain how Nanobodies can be used in breast cancer research?
Recently our group has developed a Nanobody against HER-2, human epidermal growth factor receptor 2, which is a therapeutic target in breast cancer. In about 20% of breast cancers HER2 receptors are overexpressed, altering breast cell growth and inducing uncontrolled cell division. Here we developed an anti-HER2 nanobody labelled with 68Ga for PET imaging and its 177Lu labelled version for targeted radionuclide therapy.
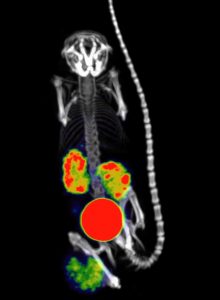 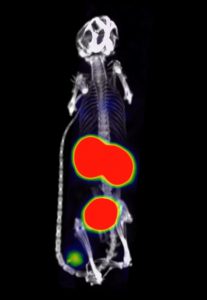 |
| Pinhole PET/CT image of 68Ga-anti-CD20 nanobody (left) and Pinhole SPECT/CT image of 177Lu-anti-CD20 nanobody made on the MILabs VECTor/CT system (Ahmet Krasniqi et al, presented at EMIM 2016) |
What is the added value of the MILabs VECTor/CT imaging system for your current research?
Currently the MILabs VECTor /CT system is utilized for more than 80 % for our preclinical in vivo screening program for Nanobodies for clinical translation and the remaining 20 % is allocated for basic research. The screening program requires a reliable system which is suitable for upto 15 scans per day with barely any need for recalibration. The MILabs system has proved to be fast, extremely stable and provides highly quantitative data, essential to our research. The ability to image both SPECT and PET radio ligands with sub-mm resolutions makes it possible to study processes inside tumors or other sub-organ structures in a very efficient way.
