Development of new oncolytic viral therapies with NIS reporter genes and high resolution SPECT
| Kah-Whye Peng, Ph.D. and Stephen J. Russell, M.D., Ph.D., are professors at Mayo Clinic and co-founders of Imanis Life Sciences (www.imanislife.com), provider of reporter gene imaging reagents and services supporting drug development research. Radionuclide Sodium/ iodide Symporters (NIS) are ideal for longitudinal 3-D mapping of biological products and processes in living animals. Imanis services include study design, image analysis, customizing lab reagents, and full-service contract research. We had the opportunity to interview these excellent scientists, in particular about novel NIS imaging technology and the benefits of ultra-sensitive and sub-half-mm SPECT. |  In 1998, Dr. Stephen J. Russell and Dr. Kah-Whye Peng joined Mayo Clinic in Rochester. They built the Department of Molecular Medicine. Their research interests include development of technologies for targeting, monitoring and enhancing the potency of novel cell, gene and virus-based anticancer therapies and to advance the most promising technologies to clinical testing. In 1998, Dr. Stephen J. Russell and Dr. Kah-Whye Peng joined Mayo Clinic in Rochester. They built the Department of Molecular Medicine. Their research interests include development of technologies for targeting, monitoring and enhancing the potency of novel cell, gene and virus-based anticancer therapies and to advance the most promising technologies to clinical testing. |
Can you explain what NIS reporter genes are and how they can be used in cancer therapy?
“NIS stands for Sodium/iodide Symporter, an endogenously expressed protein in the thyroid, salivary glands, gastric mucosa, and mammary glands of mammals; from mice to humans (and everything in between). Its physiological function is to concentrate iodine in the circulation for the synthesis of thyroid hormones”. For more than 70 years, NIS has been exploited in the nuclear medicine imaging to provide a diagnosis of thyroid disorders, with radioiodine I-123 or pertechnetate 99m-TcO4 (gamma-emitting isotopes) or therapy using radioiodine I-131 (a beta-emitting isotope) to ablate hyperactive thyroids“ explains Dr. Russell.
So how can NIS reporter genes been used for other non-thyroid oncology therapy?
“Oncolytic virotherapy is a novel therapeutic approach which uses the destructive power of viruses to selectively infect and kill target tumor cells. The therapeutic virus has the unique capability to replicate and thus spread to distant disease sites, and can be an alternative to or used in combination with traditional chemo- and radiotherapies. By selectively modifying viruses, tumor specificity and efficacy can be increased. However, the key challenge remains to understand the underlying mechanisms of vector amplification and spread within diseased tissue. The NIS reporter gene normally functions to concentrate iodide into follicular cells of the human thyroid during production of thyroid hormone, but when expressed from the genome of an oncolytic virus NIS can empower noninvasive imaging of virus replication. In addition, NIS expression in combination with radioiodine can induce synergistic tumor cell killing by concentrating high-energy isotope within infected tumor tissue“ explains Dr. Peng.
Which non-thyroid oncology treatment are you currently developing?
“Our laboratories have been using a wide array of oncolytic viruses including a modified measles virus strain (Edmonston) that has been engineered to express the NIS gene (MV-NIS) which has shown preclinical activity against a large number of oncology targets and is currently in clinical testing for the treatment of Multiple Myeloma and Ovarian Cancer.
Multiple Myeloma (MM) is a currently incurable malignancy of terminally differentiated plasma cells. Initial therapy for MM patients is with alkylating drugs, corticosteroids, proteasome inhibitors, and immunomodulatory drugs, with the addition of myeloablative chemotherapy and autologous stem cell transplantation for patients under the age of 70 years. Unfortunately, relapse is inevitable in most patients and the systemic nature of this disease limits the use of traditional radiotherapies. Another non-thyroid target is ovarian cancer, which is the second most common malignancy of the female genital tract in the United States. Despite debulking surgery and chemotherapy, more than 65% of the patients will relapse and no curative treatment option is available. These are just two examples of non-thyroid cancers in our continued attempt to use oncolytic viruses to improve the clinical outcome of these patient groups“ continues Dr. Russell .
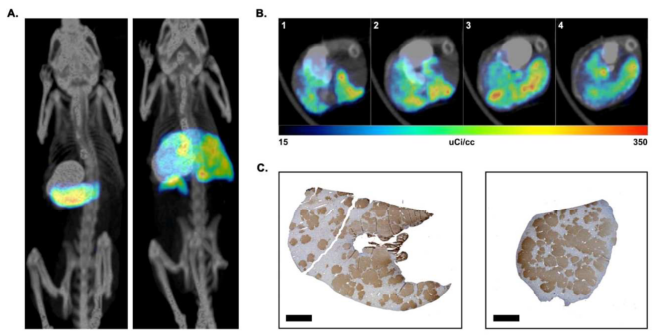 |
| In vivo imaging of NIS-labeled hepatocytes. (A) Representative fused Maximum Intensity Images (MIP) images of control hepatocyte injected (left) and NIS-labeled hepatocyte injected (right) mice. The location of the stomach was identified by oral administration of barium sulfate prior to imaging. The signals from the thyroid and bladder have been removed from both animals. (B) Representative transverse SPECT/CT sections of a NIS-labeled hepatocyte transplanted liver, showing different positions of the liver from head [1] to feet [4] of animal. (C) FAH-immunohistochemistry of the same liver showing non-uniform distribution of transplanted cells in two different lobes from the same mouse. FAH-positive cells are stained brown. Scale bar = 2mm. From Hickey et al., Liver Transplantation 2014 |
In your research which imaging tools do you use to detect and quantify the viral oncolytic activity?
“In our laboratory, we have set up a wide array of assays to detect and quantify the oncolytic activity of NIS-expressing viruses from several families in relevant in vivo cancer models. Traditionally these studies have relied on immunohistochemical staining of explanted tumors to detect the presence of virus replication, requiring a lot of animals. Recently, we have been using the MILabs U-SPECT/CT system which allows us to perform high-resolution NIS reporter gene imaging studies for detection of intratumoral viral spread and viral oncolytic activity in 3D, and quantification in vivo during follow-up of the animals” explains Dr. Peng.
What is the added value of MILabs systems for your research?
“In the past preclinical SPECT imaging was hampered by poor resolution which makes it very difficult to see the viral spread inside a tumor in a treated animal. The MILabs U-SPECT/CT system provides us extremely high-resolution SPECT images (less than 0.25 mm) so we can track the expansion of individual infectious centers of a NIS-expressing oncolytic virus in a single tumor nodule in a living animal. Moreover, the ability to quantify in vivo the oncolytic activity of our NIS reporter gene is greatly reducing the number of animals used to complete a study. Outside oncolytic virotherapy, we are finding that the technology is very versatile for application to many other research problems. For example, we have been using the MILabs U-SPECT/CT system for longitudinal in vivo analysis of the process of liver regeneration by NIS expressing hepatocytes, the biodistribution of gene delivery to various tissues by NIS-expressing AAV vectors, the responsiveness of NIS-expressing tumors to anticancer drug therapy and the progress of tissue fibrosis in NIS transgenic fibrosis reporter rats” concludes Dr. Peng.
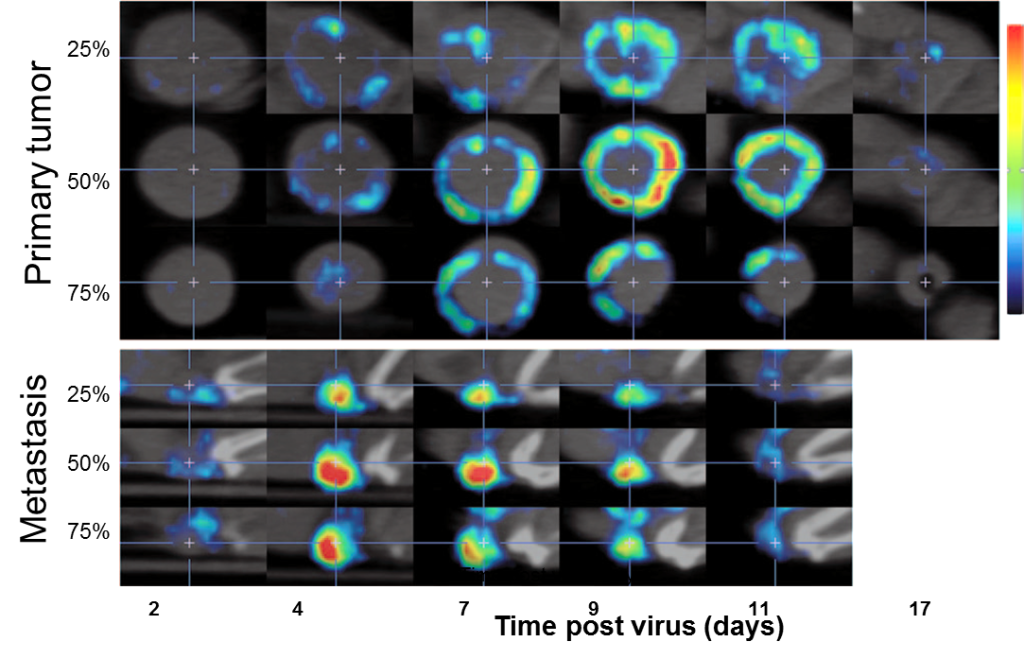 |
| Serial images in the sagittal plane of a primary tumor (top) and metastatic tumor (bottom) from the same animal at multiple time points following IV administration of MVvac2 NIS. Images from different depths within the same tumor are shown; the relative position of each image within the total tumor depth is indicated on the left. From Miest et al., Molecular Therapy, 2013 |
